Intravenous
Delivers therapies to the brain through the blood, via a cannula inserted into a vein. We use this for ATMPs that we know will cross the blood-brain barrier.
Intrathecal
Delivers therapies directly to the cerebrospinal fluid (CSF) that surrounds the brain, via a lumbar puncture, which is when a needle is inserted into the lower back, between the bones of the spine.

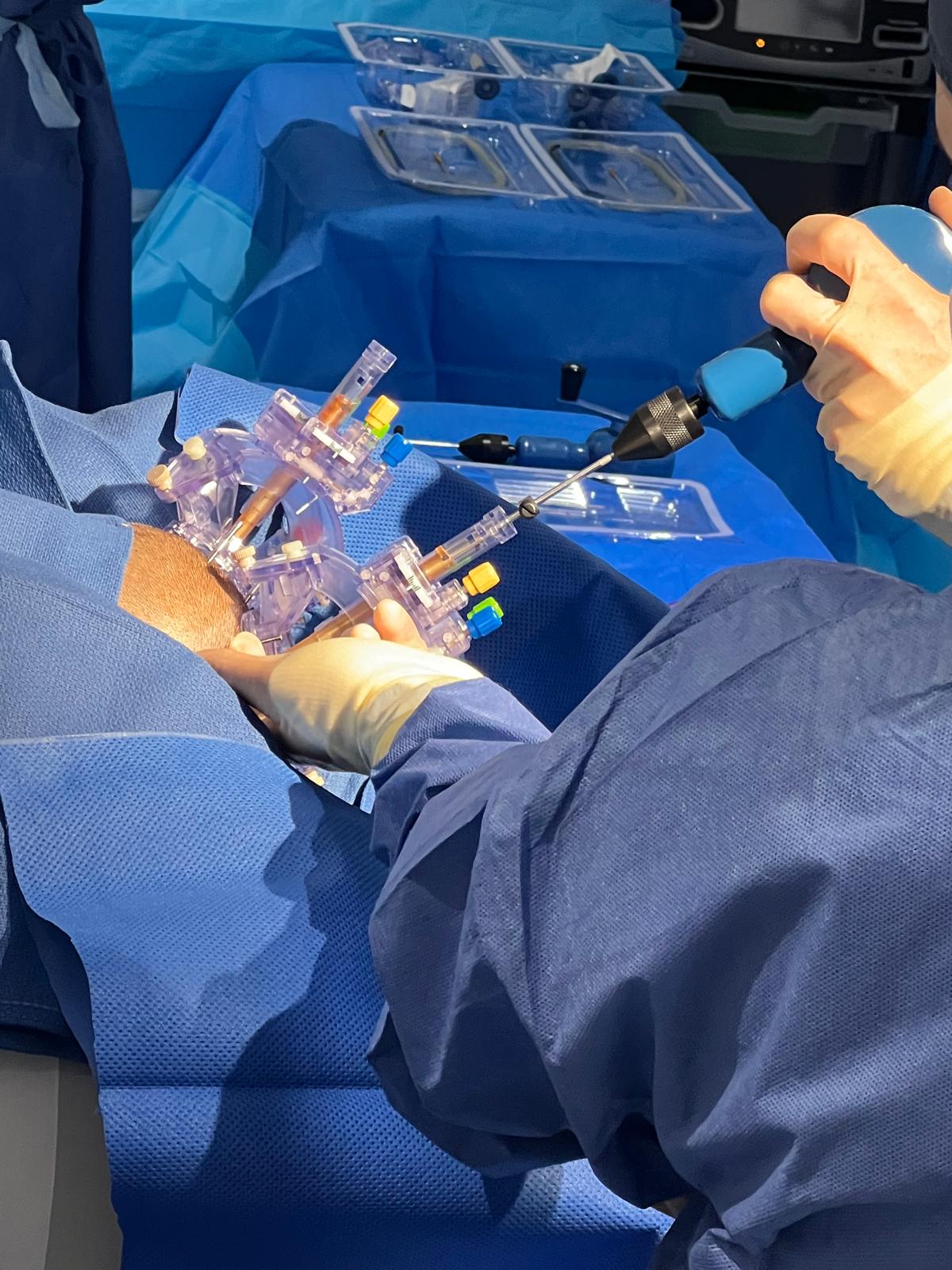
Neurosurgical
Delivers therapies directly to specific areas of the brain using surgically placed devices while the patient is in an MRI scanner, to see exactly where the therapy is going in real-time. We are the only centre in the UK (and one of just five worldwide) with the expertise and facilities to safely deliver these neurosurgical treatments.